OEM spectrometers for process analytical technology
What is Process Analytical Technology (PAT)?
Process Analytical Technology (PAT) is a framework introduced by the U.S. FDA in 2004 to improve the design, monitoring, and quality control of pharmaceutical and food production processes. The goal of PAT is to ensure consistent final product quality by understanding and controlling the manufacturing process in real time.
PAT works by monitoring and adjusting Critical Process Parameters (CPPs) to maintain the desired Critical Quality Attributes (CQAs). When CPPs are measured continuously — for example, using in line sensors — manufacturers gain deeper process understanding, reduce over processing, improve consistency, and minimize waste.
Reliable, high quality data is essential for PAT. One of the most effective tools for generating such data is the near infrared (NIR) spectrometer, which can be placed directly in the production line for continuous monitoring.
Why are Ibsen’s NIR spectrometers well suited for real time PAT monitoring?
Ibsen’s NIR spectrometers are engineered specifically for demanding PAT environments:

Why in-line monitoring?
In line monitoring provides immediate feedback, enabling manufacturers to adjust the process instantly rather than waiting for delayed off line laboratory results. This real time control improves product quality, reduces batch failures, and supports the core purpose of PAT: consistent, data driven manufacturing.
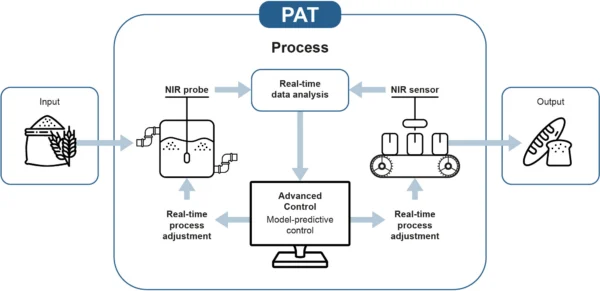
Why use NIR spectrometers for PAT?
Near infrared (NIR) spectrometers offer several advantages for PAT applications:

What specifications matter most when choosing an NIR spectrometer for PAT?

When selecting an NIR spectrometer for PAT, the most important specifications include:
Our NIR spectrometers for PAT
Our NIR spectrometers are optimized for different applications, from cost efficient, compact spectrometers for handheld devices to the highest performance spectrometers with low stray light and high signal-to-noise ratio.
For more information on our range of spectrometers for NIR absorption spectroscopy, please select your wavelength range of interest:
Wavelength ranges: NIR (900 – 2500 nm)

More resources
Want to know more?
For further information see below.
