Near-infrared spectroscopy
Near-infrared (NIR) spectroscopy is a method commonly used to analyze organic molecules in the production of pharmaceutics and food. The technology is promoted by the Food and Drug Administration (FDA) or Process Analytical Technology (PAT).
A production site will often require multiple NIR sensors and it is crucial to have transferability of results between the different sensors. Transferability means that measurements done with different instruments on the same sample gives identical results.
Ibsen’s ROCK NIR, ROCK XNIR and PEBBLE NIR spectrometers are specifically designed to have low unit-to-unit variation and thereby enable good transferability.
NIR and IR Spectroscopy
The infrared spectrum is the wavelength range from 3,000 nm to 10,000 nm whereas the near-infrared spectrum is the range from 800 – 2500 nm.
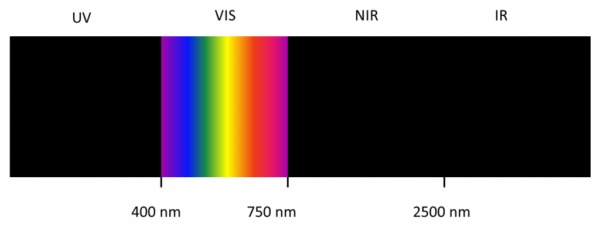
Since the energy carried by light (photons) is inversely proportional to the wavelength, IR light has low energy due to its long wavelengths. For this reason, when IR light is absorbed by a molecule the low energy is transformed into translation and bending of the molecules.
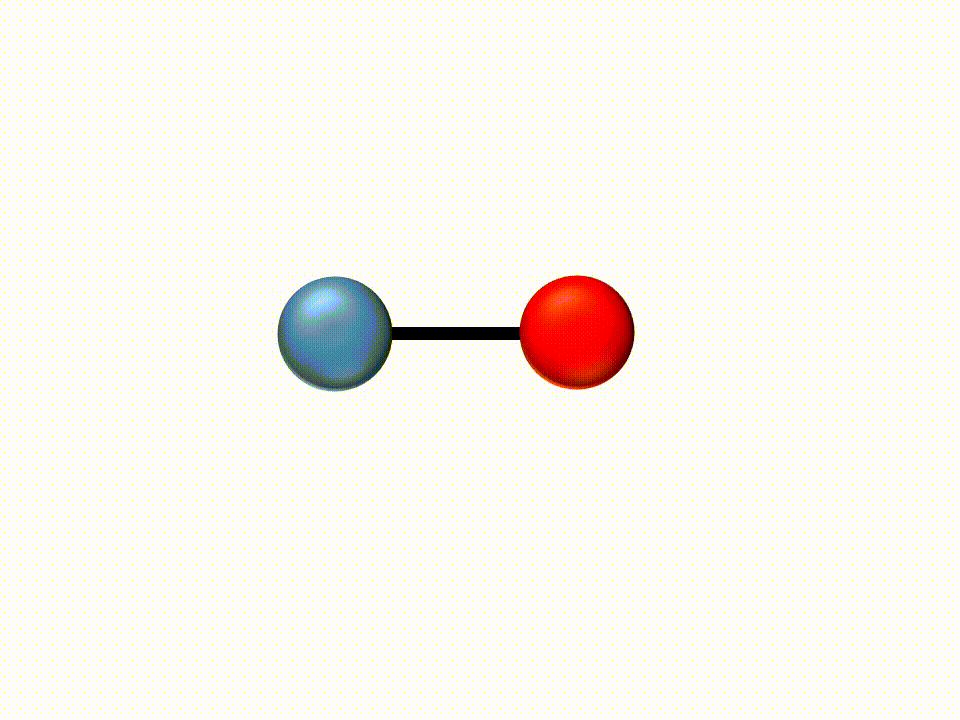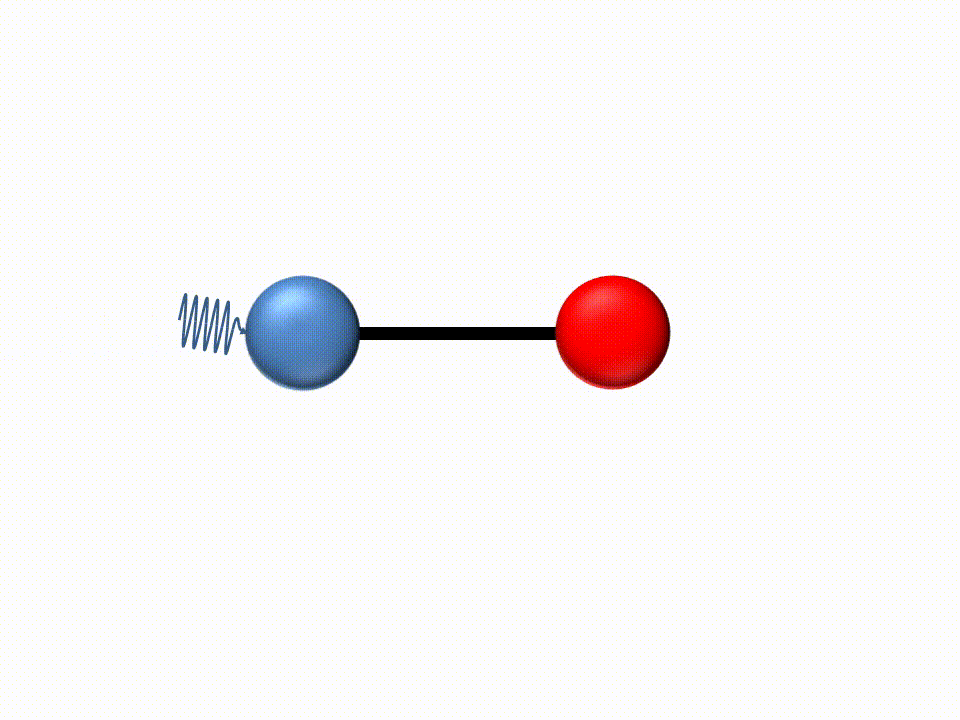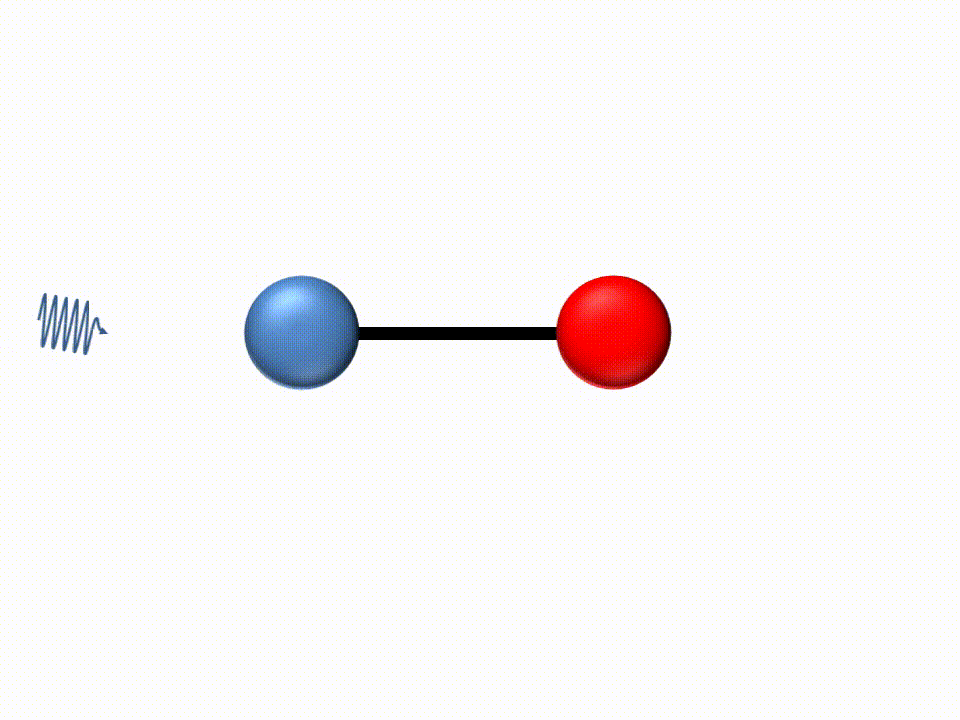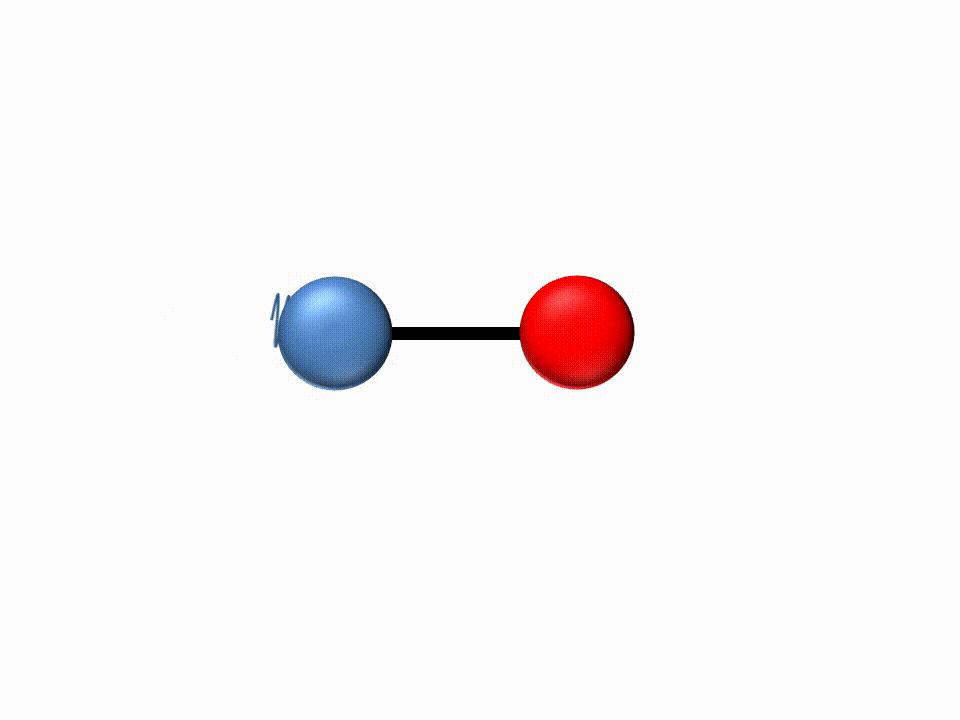
|
IR spectroscopy is thus an absorbance method where different molecular structures will absorb light at different wavelengths.
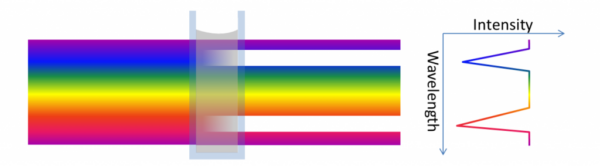
In general, IR spectroscopy provides stronger absorbance and sharper peaks than NIR spectroscopy does. However, near-infrared spectroscopy is generally preferred for PAT because NIR instruments are more stable (no moving parts) and are easier to implement as an in-line instrument on the factory floor.
| IR | NIR |
|---|---|
| 3 - 10 µm | 0.8 - 2.5 µm |
| Best performance due to strong and sharp peaks | Poorer performance due to weak and broad (overlapping) peaks |
| Issues with robustness - Requires moving parts /mirror) (FT-IR) that are sensitive to vibration | Robust against vibration - No moving parts |
| Difficult to implement as in-line method - Requires probe to touch sample (ATF) or sample to be mixed with powders | Easy to implement as inline method - No touch technology |
The complexity of near-infrared spectra
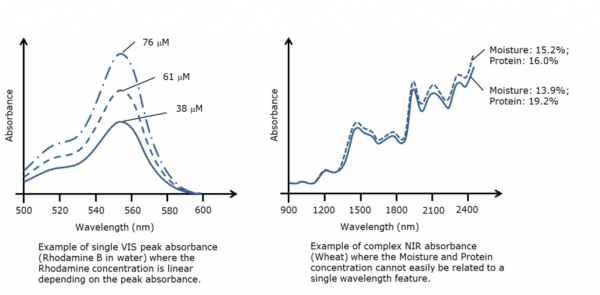
The pictures below show to the left an example of a simple single peak absorbance (Rhodamine B in water), where the concentration can be found simply by measuring the peak absorbance. To the right is a typical near-infrared spectrum (wheat) where the concentration of water and protein is very difficult to relate to a single peak in the spectrum.
Chemometrics
Due to the broad and overlapping peaks found in near-infrared spectra, advanced numerical (software) methods like Chemometrics are typically used to extract parameters like concentrations from the spectral data. These methods are quite complex and typically builds on multi-variate statistical methods.
The overall process used in chemometrics is shown below:
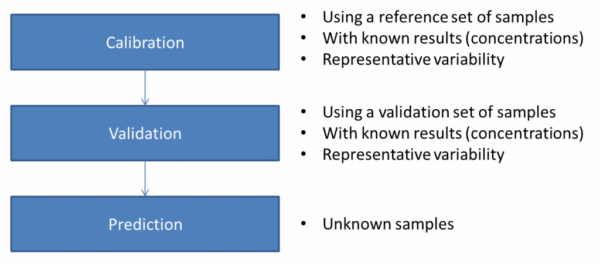
The calibration step is where you build your model from a large set of NIR spectra measured on samples with different but known concentrations. A common method used in Principal Components Analysis (PCA) which takes the 100s of correlated spectral data (intensity versus wavelength) and reduces it to a small set of new independent variables – called Principal Components (PC).
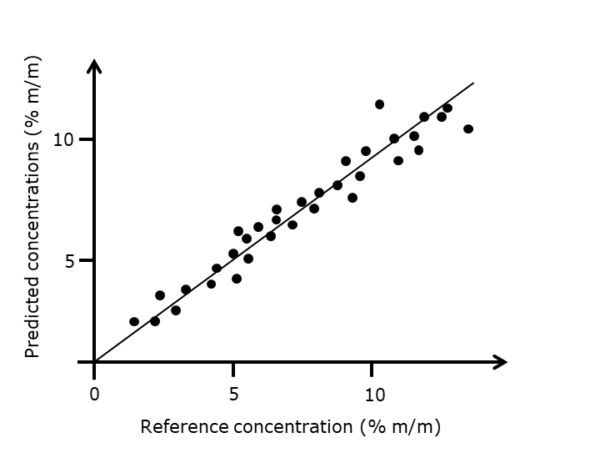
In the validation step, you test how well your model can predict the correct concentrations of a known set of samples. The figure below shows a typical example of a model that provides a fairly linear relation between the known reference concentrations and the concentrations predicted by the chemometric model. Thus, we may use this model to predict concentrations based on near-infrared spectra measured on unknown samples.
Products for NIR spectroscopy
ROCK XNIR
ROCK XNIR Home / Products / OEM Spectrometer [...]
PEBBLE NIR
PEBBLE NIR Home / Products / OEM spectrometer [...]
Want to know more?
For further information see below.

